Zoe Martin, Policy Manager at Cancer Research UK, gives an overview of their joint report on UK-EU scientific collaboration.
Pinpointing how UK-EU collaboration advances medical research
14 Aug 2017
CaSE readers shouldn’t be surprised to hear that international collaboration is good for science. Many of you will regularly work with scientists in other countries and be part of diverse international teams. Collaboration is so fundamental that the new model biomedical research facility – the Francis Crick Institute – has laboratory and office areas designed around central collaboration spaces.
Demonstrating joint working, Cancer Research UK partnered with seven other leading UK medical research funders to investigate the impact of UK-EU scientific collaboration. The resulting report, aimed at informing EU and UK based audiences during Brexit negotiations, identifies how the UK has contributed to EU science, and ultimately patient health across the EU.
The main message is clear: scientists have a greater impact when they collaborate. The UK has been a major contributor to EU programmes, cited as an important instigator and co-ordinator. The findings provide a bank of evidence to help make the case for continued collaboration in research as we leave the EU – one of Cancer Research UK’s priorities for the new Government.
Five areas the UK was recognised to have made key contributions were:
1. EU collaborations, advisory bodies and policies
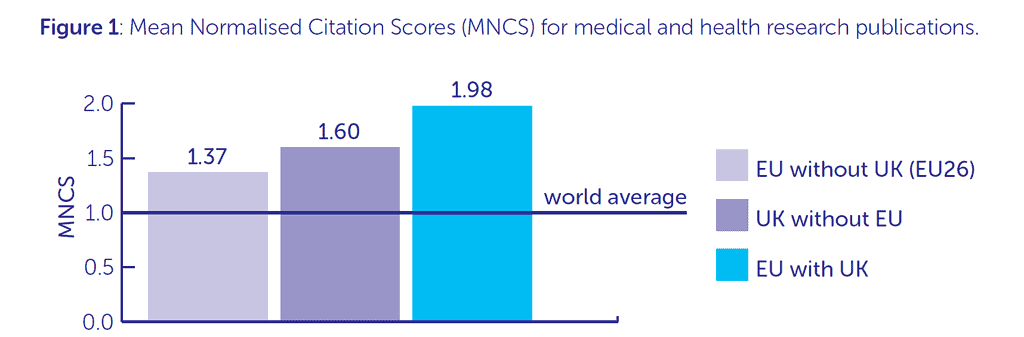
The UK, with its diverse research ecosystem, is a key scientific partner in the EU. UK-based medical researchers’ most common collaborations are with European researchers (18% of total). The graph below shows how research publication citation scores reach almost double the world average when the UK and EU work together. UK input into EU committees and advisory bodies was also highly valued, as well as our contribution to regulatory and ethical expertise.
2. Development of pan-EU clinical trials
The UK is established, active and respected when it comes to clinical trials. We run more phase I trials than any other EU nation, and the second highest number of phase II and II trials. The UK and EU work extensively as partners, totalling over 4,800 joint UK-EU clinical trials between 2004 and 2016. Of the 200 clinical trials directly funded by Cancer Research UK, 28% involve patients from another EU country.
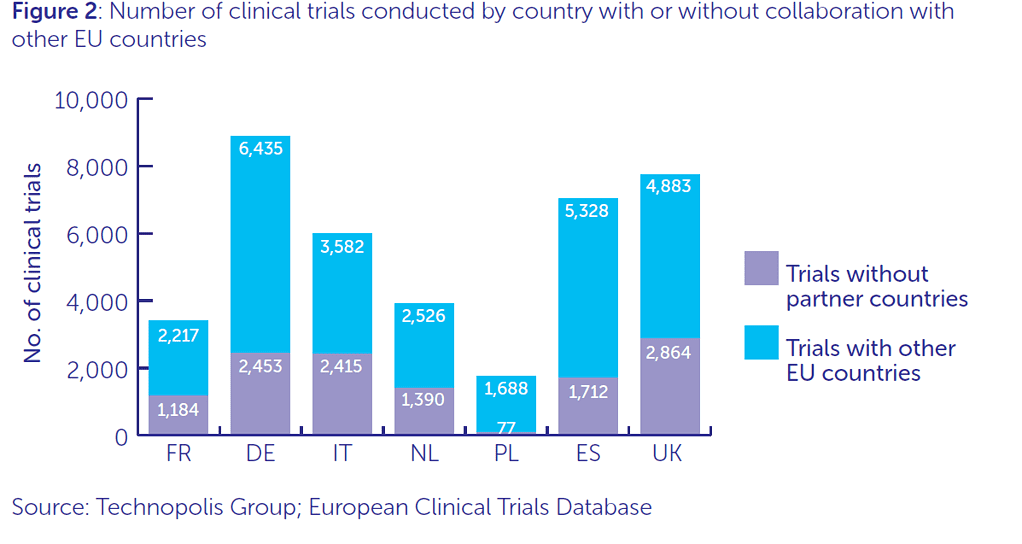
Pan-EU collaboration is particularly important in rare diseases and paediatrics where patient numbers may be too small for single country trials. To develop better interventions for these types of disease, the UK must continue to be able to run trials with other countries
3. World-class medical research facilities
The UK has excellent medical research facilities. Examples include the bioinformatics centre at the Wellcome Trust Sanger Institute, and bio-repositories like the Public Health England Culture Collections. These collections are accessed by scientists from across the globe, so directly enable research collaboration.
4. Development of new therapies and medical technologies
The UK’s record in translational research is impressive, having discovered a quarter of the top 100 prescription drugs worldwide. UK research also contributes to clinical guidelines in multiple EU member states and disease areas.
The UK has also championed patient involvement with the report showcasing a case study on person-centred dementia care. Here, “well-being” was incorporated as a success indicator in UK dementia care before gradually being adopted across the EU.
5. An attractive training environment for researchers
Underpinning scientific advances is a highly skilled research workforce, which the UK plays a pivotal role in training. Around 16,000 students from other EU countries are registered on UK biomedical courses, including 6,500 postgraduates. The UK is described as empowering graduates to think analytically and innovatively. Global mobility is a key feature of the UK medical research community and beneficial to scientific progress.
What next?
As the UK develops a new relationship with the EU it is vital that science and patients are prioritised in negotiations. Cancer Research UK is calling for the Government to ensure that:
- Our immigration system allows us to continue to attract and retain a pipeline of scientific talent from across the globe.
- We are aligned to EU legislation to enable us to continue to collaborate on clinical trials.
- Patients are able to access the drugs they need through an aligned approach to the marketing authorisation and licensing of drugs. It is vital the UK remains an attractive place to launch innovative treatments.
- The UK continues to have access to EU funding programmes and play a role in their development.
Details set out in future arrangements will be key. Any limitations on the ability of researchers and institutions to work together could diminish the impact of science both in the UK and the EU.
The study was conducted by Technopolis Group and co-funded by the Academy of Medical Science, Arthritis Research UK, the Association of Medical Research Charities, British Heart Foundation, Cancer Research UK, Medical Research Council, MQ: Transforming Mental Health and Wellcome.
Related articles

The Physiological Society’s policy team on the health challenges facing older workers and the urgent need to develop a strategy to ensure older people are happy and healthy at work.

Jo Reynolds, Director of Science and Communities at the Royal Society of Chemistry, on the RSC’s new summary report looking to unlock the potential of deep tech SMEs.

Lisa Morrison Coulthard, Research Director at the National Foundation for Education Research, on the Nuffield Foundation funded five year research programme providing insights into the essential employment skills needed for the future workforce

Sir Adrian Smith, Institute Director and Chief Executive of The Alan Turing Institute, and Graeme Reid, Professor of Science and Research Policy at UCL, set out the findings from their new independent report on international partnership opportunities for UK research and innovation